..says drug contains iron wires .
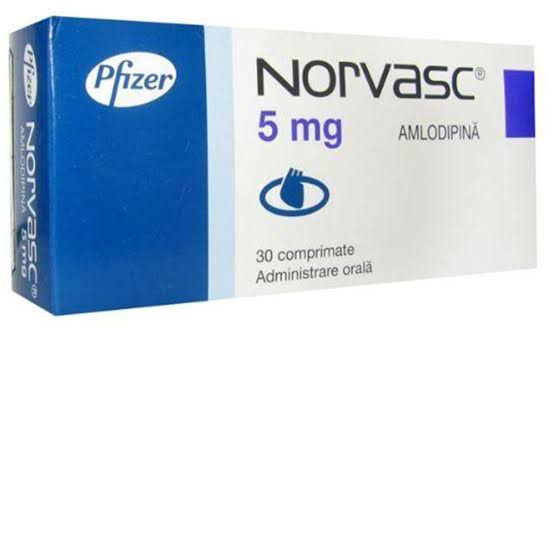
The National Agency for Food and Drug Administration and Control ( NAFDAC) has warned against the use of the drug, Norvasc 5 mg
In a statement, the Edo state Commissioner for Health,Prof. Obehi Akoria said that “the Taiwan Food and Drug Administration ( FDA) has ordered the recall of six million tablets from two batches of Norvasc 5 mg tablets produced by US based drug developer,Viatris Inc, following the discovery of iron wires in samples taken from shipment.
According to the statement, the discovery” makes the two batches of Norvasc 5 mg tablets substandard”.

Share this:
- Share on X (Opens in new window) X
- Share on Facebook (Opens in new window) Facebook
- Share on WhatsApp (Opens in new window) WhatsApp
- Share on Pinterest (Opens in new window) Pinterest
- Share on Telegram (Opens in new window) Telegram
- Share on Tumblr (Opens in new window) Tumblr
- Share on LinkedIn (Opens in new window) LinkedIn
- Share on Reddit (Opens in new window) Reddit
Related
Related posts
0
0
votes
Article Rating
Subscribe
Login
0 Comments
Oldest
Newest
Most Voted
Inline Feedbacks
View all comments
Recent Posts
House of Reps prescribes 2-year jail term, 10m fine for dual party membership
Share this… Pinterest Facebook Twitter Linkedin Wechat Whatsapp Telegram Blogger The House of Representatives has amended Section 77 of the recently assented Electoral Act 2026 to prescribe two-year jail term or N10 million fines or both, for anyone who knowingly maintains membership of two political...
After going through most rigorous Screening,”It’s enough to say you are fit and ready to deliver your best”Gov Fubara To New Commissioners
Share this… Pinterest Facebook Twitter Linkedin Wechat Whatsapp Telegram Blogger Rivers State Governor,Sir Siminialayi Fubara has described the screening exercise on the new commissioners as one of the most rigorous. Speaking during the inauguration of five of the commissioners confirmed by the State House of...

